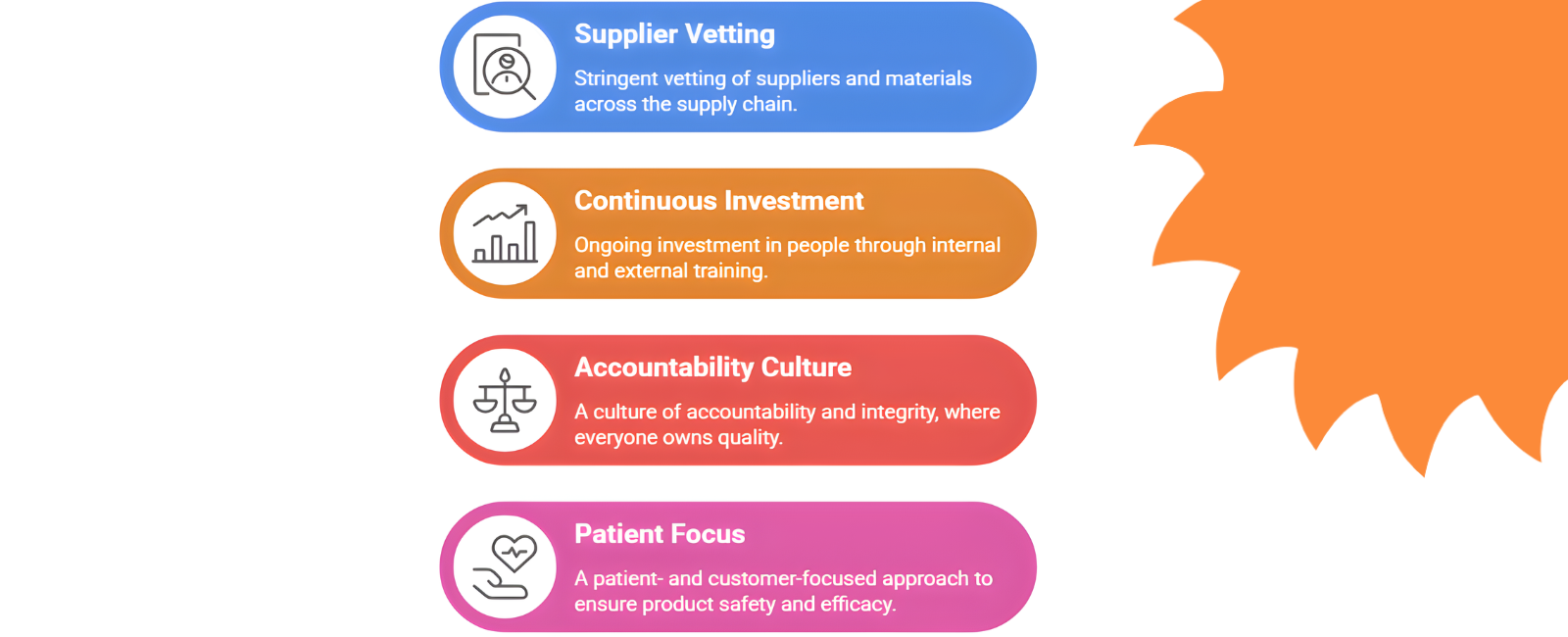
“At Curexa, every connection we build is driven by purpose — to bring authentic, high-quality healthcare within everyone’s reach. The Commercial team is the pulse of Curexa’s growth, turning insight into opportunity and relationships into results. We lead with strategy, creativity, and conviction, ensuring our therapies not only reach the market but also earn lasting trust. Guided by our values of Integrity, Mindfulness, Passion, Agility, Collaboration, and Trust (IMPACT), we champion Curexa’s vision across every channel and partnership. For us, it’s not just about market share — it’s about shaping a brand that stands for care, credibility, and impact.”